What is HHO?
What Is HHO Gas? A Beginner’s Guide to Oxyhydrogen Technology
HHO gas, also known as oxyhydrogen, is a powerful and clean-burning fuel made by splitting water (H₂O) into two parts of hydrogen and one part oxygen. This 2:1 mixture creates a highly reactive gas with 99.95% purity, often referred to as Bang Gas or Knallgas in German.
In this article, you’ll discover how HHO gas is produced, how it works, and why it’s gaining popularity in automotive and industrial applications.
How Is HHO Gas Made?
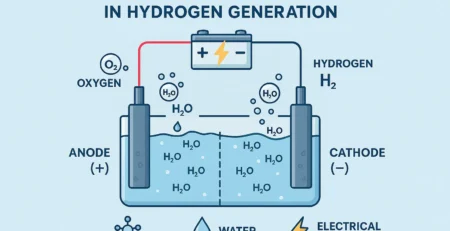
The most common way to produce HHO gas is through a process called electrolysis. First discovered by Michael Faraday in 1832, electrolysis uses electricity to separate water into its base elements: hydrogen and oxygen.
During electrolysis:
-
A direct current (DC) flows through water mixed with an electrolyte (like KOH or NaOH).
-
This current splits the water molecules.
-
The result is a highly combustible mix of HHO gas.
Although electrolysis is the preferred method, there are other ways to produce hydrogen. However, they are usually more complex or less energy-efficient.
Why Is HHO Gas Explosive?
Because HHO gas contains both fuel (hydrogen) and oxidizer (oxygen) in perfect ratio, it ignites instantly when exposed to a spark. Even at just 4% concentration in air, it can cause an explosion. Therefore, handling HHO gas requires extreme care, especially under pressure.
What Are the Uses of HHO Gas?
HHO gas is incredibly versatile. Here are the most common uses:
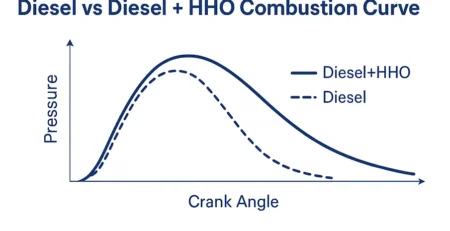
✅ 1. Automotive Engine Carbon Cleaning
Many people install HHO kits in cars and trucks to:
-
Reduce fuel consumption
-
Lower emissions
-
Clean carbon deposits from the engine
The gas helps burn fuel more completely, which leads to better performance and cleaner engines.
✅ 2. Welding and Cutting
HHO torches are used for:
-
Metal cutting and welding
-
Engraving and polishing
-
Electronic component work
Because HHO burns at high temperatures, it’s ideal for precision metalwork.
✅ 3. Industrial Applications
Industries use electrolysis and HHO gas for:
-
Electrometallurgy (extracting pure metals like aluminum, sodium, and lithium)
-
Producing chlorine and sodium hydroxide
-
Electrofluorination for chemicals like trifluoroacetic acid
These applications benefit from the clean and efficient energy that HHO provides.
✅ 4. Space and Submarine Systems
Electrolysis systems on spacecraft and nuclear submarines produce oxygen for breathing and hydrogen for energy.
✅ 5. Artifact and Metal Restoration
Electrolysis is used to clean and preserve old metal objects. You can restore items like:
-
Rusty coins
-
Engine parts
-
Iron tools
With just a bucket, battery charger, and some basic materials, anyone can use electrolysis in a home workshop.
How Safe Is HHO Gas?
Although HHO gas is efficient and eco-friendly, it’s highly flammable. Never compress HHO gas or let it accumulate in enclosed spaces. Always follow proper safety protocols when using or generating HHO.
Conclusion: Why Learn About HHO Gas?
HHO gas offers a compelling alternative to traditional fuels. It’s clean, efficient, and can be generated on-demand from water. Whether you want to lower your vehicle emissions, restore old parts, or explore hydrogen-based technologies, understanding HHO gas is a great place to start.
As global industries seek sustainable energy solutions, oxyhydrogen technology stands out as a powerful tool for the future.
Subscribe our Newsletter